In the late 1980s, the aptly named Package Products Company in Charlotte, North Carolina, was facing a toxic problem. The consumer packaging company printed eye-catching candy wrappers, soft-drink shrink wrap, and other materials doused with colorful inks. But the company discovered that the chemicals in the ink vaporized when the wrappers were set out to dry, poisoning the air and creating a dangerous environment for workers and even those who lived nearby.
The company brought in Michael Lukey, an air pollution control engineer, to solve the problem. He had a novel idea: He set up blowers that pumped clean air into the room that contained the printing press. As the air traveled through the area, it swept up the volatile chemicals emanating from the ink. On the other end of the room, Lukey used exhaust fans to funnel the chemical-laden air out of the room and into equipment that incinerated the toxins.
The plan worked as Lukey had hoped. Once the design was operational, the company concluded that the technology neutralized more than 97 percent of the chemicals. Since the method involved functionally sealing off the room in order to use air flow to isolate toxins, it became known as a Permanent Total Enclosure, or PTE.
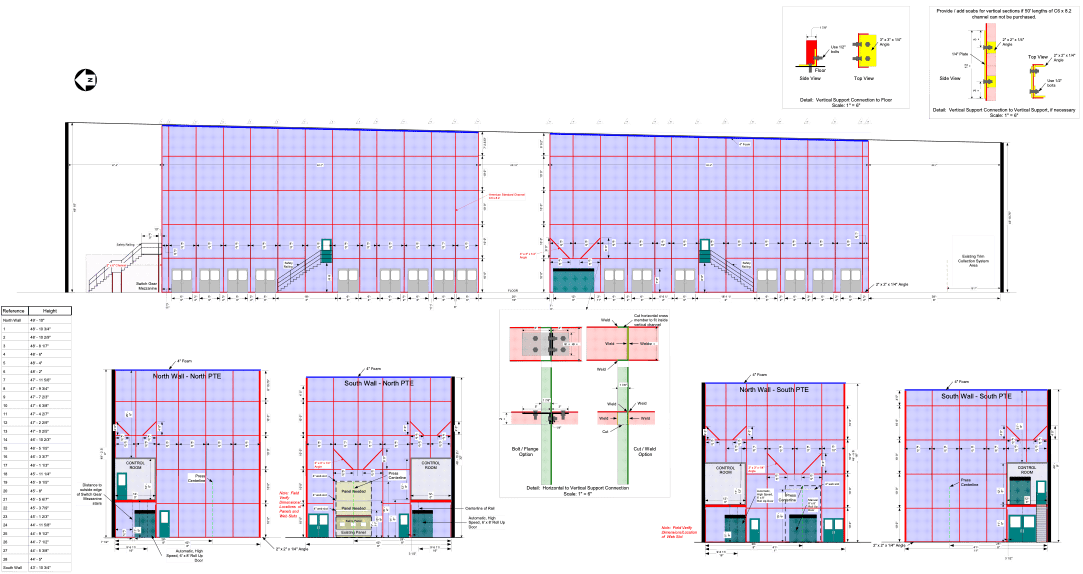
Within a couple of years, the Environmental Protection Agency had expressed interest in the technology. “The EPA got wind of the kind of facility that we put in over there and wanted to hear more about it,” Lukey told Grist. “They initiated some studies and did comment on the appropriateness of these kinds of larger enclosures that would reduce volatile organic chemical emissions from these printing operations.”
The EPA would ultimately decree PTEs the “Best Demonstrated Control Technology” for reducing harmful emissions from operations like Package Products Company’s, and the technology spread across the country. From shipping facilities to spray-painting booths at car manufacturers, PTEs became a go-to pollution solution in a handful of niche industries. In his decades on the job, Lukey has personally designed more than 300 PTEs. He thinks he’s probably worked for every candy wrapper manufacturer in the country, including a company that produces the iconic bright yellow M&M’s wrapper.
The EPA is now betting on PTEs to solve a much thornier problem: Roughly half of all medical devices in the U.S. are sterilized with ethylene oxide, a profoundly toxic chemical that the EPA has determined can cause multiple forms of cancer. When companies fumigate medical devices with ethylene oxide, it often leaks out of their sterilization plants, poisoning workers and community members nearby. In 2016, the EPA found that the chemical was 30 times more toxic to adults and 60 times more toxic to children than previously understood. Two years later, the agency issued alerts to residents living by sterilizers with unusually high cancer risk, prompting local advocates to file lawsuits against the facilities and pressuring lawmakers and regulators to shut them down. Earlier this year, in response to a court-imposed deadline, the EPA finally issued regulations intended to bring down emissions of the chemical.
The new rule relies on PTEs to safeguard exposed communities. But engineers, former EPA officials, and air quality experts told Grist that, while PTEs are effective at mitigating pollution from equipment in small, enclosed rooms, PTEs have rarely been tested at facilities on the scale at which medical device sterilization is conducted. And in the few cases where PTEs have so far been installed at sterilizer facilities, while ethylene oxide levels decreased overall, some still breached federal safety thresholds when measured by nearby air monitors. The problem comes down to basic engineering: The spray-painting booths that PTEs typically treat tend to be about the size of the average American living room. Sterilization facilities, on the other hand, can occupy tens of thousands of square feet.

“EPA has not one shred of engineering analysis to show it will work,” said Ron Sahu, a mechanical engineer and consultant with decades of experience working with state and federal environmental regulators and industrial companies. “It is a faith-based, hopeful suggestion, and we’ll see how it works.” Sahu has worked as a court-approved technical expert in litigation against the sterilization industry and has submitted reports to the EPA on the efficacy of PTEs on behalf of environmental groups.
Using the technology in large warehouse-like structures with multiple entry points and windows makes it nearly impossible to ensure that air tainted with ethylene oxide doesn’t escape from the facility before it can be treated, experts told Grist. While the EPA is requiring companies to continuously monitor air flow or pressure to ensure PTEs work as intended, facilities are allowed to average data over three hours — an interval that experts told Grist will not guarantee that PTEs are capturing all ethylene oxide emissions.
“It’s very difficult to have a warehouse-sized PTE,” said Jonathan Thornburg, director of exposure and aerosol technology at the nonprofit Research Triangle Institute in North Carolina. He added that even “the wind blowing against the side of the building can change the pressure differential” and allow contaminated air to escape.
“We strongly disagree with the assertion that PTE is not appropriate to apply to this source category,” Angela Hackel, an EPA spokesperson, told Grist, adding that “properly designed and operated” PTEs can capture all ethylene oxide emissions. The agency’s modeling shows the technology can reduce excess cancer risk to acceptable levels, she said, and that it has been “applied widely to any industrial processes that need to control” emissions of volatile chemicals.
If these measures do not work as intended, however, the EPA is in a bind, in large part because banning ethylene oxide use is not an option. Medical devices must be sterilized before they can be safely used in hospitals, and some can only be sterilized with ethylene oxide. Since alternatives don’t yet exist, the EPA cannot require the sterilization industry to phase out the chemical. In the meantime, the agency’s approach has been to mandate that facilities reduce its use to the lowest extent possible without compromising patient safety — and to install PTEs, which it has selected as an effective solution for this growing problem.
But the sterilization industry itself is crying foul over this approach: The country’s major sterilizers have already informed the EPA that strict compliance with PTE guidance is not feasible. In instances where companies have installed the technology, it has failed to reduce ethylene oxide emissions to safe levels. In Southern California, for instance, local regulators forced Parter Medical Products to shut down in 2022 after an air quality monitor near the facility detected ethylene oxide levels more than 4,000 times above the EPA’s safe limit. After installing a PTE, emissions decreased, but the company still exceeded public health thresholds. The current emissions from the facility put the cancer risk for nearby residents at 378 in 10,000 — 378 times above the EPA’s threshold. Similarly, after a PTE was installed at a Medline facility in Illinois, regulators found ethylene oxide emissions more than 45 times above the EPA’s threshold.
In an email, an EPA spokesperson downplayed these numbers, saying that sampling the air for ethylene oxide can often yield inaccurate results, since other compounds in the air can cause concentrations to appear higher than they actually are.
“This is why we use modeling data as our driver which we believe to be more accurate,” she said, adding that agency models had found the maximum risk to communities from sterilizers after the installation of PTEs would not exceed federal standards. But state and federal regulators have used ethylene oxide monitors on various occasions to gauge a community’s risk of exposure to the compound. In fact, EPA and state regulators have used monitoring data to shut facilities down and require the installation of additional control technology.

In order for a PTE to comply with EPA standards, it needs to meet certain criteria. All doors, windows, and openings need to be closed during operations and should be limited in size. The air should flow into the structure at a certain velocity. If these criteria are met after the PTE is installed and when regulatory agencies conduct inspections, the technology is considered 100 percent efficient. That is, it’s assumed that the PTE is capturing all the pollution in the air.
These criteria will be difficult to meet, given that the typical sterilization facility has a number of openings to the outside: Massive garage doors open and close multiple times a day when trucks stop to deliver unsterilized products and pick up equipment after it’s been treated. As a result, maintaining an air barrier such that the ethylene oxide inside the facility doesn’t escape is a challenge with no obvious solution, engineering experts told Grist. Additionally, without requirements to continuously monitor the air quality around a facility, the EPA and local regulatory agencies have no way of knowing if a PTE is working as intended.
“What is required is that they meet the criteria, but it’s ambiguous and left to the client how they prove compliance,” said Lukey. “The onus for compliance is on the company.”

In order to ensure facilities are complying with the requirements year-round, companies need to do weekly inspections, Lukey said. Doors and windows need to be checked regularly to ensure they remain closed. The velocity of the air flowing through the room where sterilization occurs needs to be monitored. And companies need a maintenance log to ensure repairs occur on a regular basis. If these types of checks don’t occur routinely, the PTE could malfunction. Lukey recalled one instance in which a client in a different industry installed 15 PTEs in a facility. When Lukey visited the premises last year, he found that half of them were not working properly.
“Companies have to have an interest in this as well,” he told Grist. “Just putting it there and leaving it doesn’t necessarily constitute continuous compliance.”
Continuous compliance is particularly important to ensure that polluted air doesn’t leak from the facility. The EPA’s criteria require that companies maintain a certain pressure differential between the inside and outside of the facility. Air must be continuously blown into the facility at a specific rate and only exit through exhaust fans that are hooked up to an incinerator that can burn the ethylene oxide. The rates at which air is fed and removed from the enclosure determines the pressure difference. The EPA requires that the difference be at least 0.00025 pounds per square inch — a tiny fraction of air pressure at sea level, which is 14.7 pounds per square inch. The difference is so small that it can be subverted by gusty winds or a cracked window.
In public comments submitted to the EPA, sterilization companies have stated bluntly that it is not possible to install PTEs that meet the agency’s criteria. Specifically, they have pointed to the fact that sterilization facilities have numerous openings to the outside and that it is not feasible to ensure air doesn’t escape through them year-round.
“The use of a PTE has not been established as a proven control technology for existing sterilization operations and is not compatible and sustainable for routine application in this context,” one company wrote. “Although these PTE requirements may be compatible with some sterilization facilities, they were developed for paint booths or other single-room operations and are not transferrable to the entire commercial sterilization source category,” according to another.
“There is no ‘one-size-fits-all’ approach for the medical technology industry,” a spokesperson for the medical technology industry group AdvaMed wrote in an email to Grist. “PTE would have low effectiveness in reduction of EtO [ethylene oxide] emissions where there are already areas of low EtO concentration. Even before the rule was finalized, sterilizers have undertaken extensive efforts to implement state-of-the-art upgrades allowing for continued safe use of EtO in order to meet and even exceed regulations.”
The EPA is requiring that facilities continuously monitor the difference in pressure inside and outside the facility, or the rate of airflow through the facility, in order to prove that the PTE is working as intended. When an agency inspector visits, they could review the company’s monitoring logs and verify whether the appropriate metrics were maintained throughout the year.
That these measurements are reported as a three-hour rolling average instead of a continuous stream of data implies that fluctuations in the pressure or flow of air may be hard to catch. Hackel, the EPA spokesperson, defended this approach, saying that it would provide data “representative of the actual operation of the permanent total enclosure.” Sahu, the engineering expert, said that averaging data will not ensure complete capture of ethylene oxide emissions.
“The devil’s in the details, and the EPA doesn’t have the details,” he said.


